Concept 34.5: Neurons Are Organized into Nervous Systems
A nervous system consists of many neurons, interacting with each other by way of synapses. When we say “many,” we mean a huge number. The human brain is currently estimated to contain about 86 billion neurons!
The simplest nervous systems seen in animals alive today occur in anemones, corals, jellyfish, and other cnidarians. In these animals, the individual neurons are widely and relatively randomly dispersed throughout the body. Together they form a nerve net:
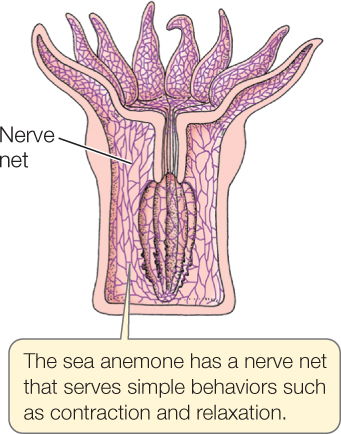
Biologists generally assume that when nervous systems first evolved they were similar to nerve nets.
As nervous systems evolved further, they followed two major trends, centralization and cephalization. Some neurons became specialized to integrate the signals of other neurons. Most of these integrating neurons became clustered together in centralized integrating organs, rather than being distributed randomly throughout the body. Centralization refers to this evolutionary trend toward clustering. At the same time, the major integrating areas became concentrated toward the anterior end of the animal’s body, a trend called cephalization (from a Greek word for “head”). Mammals vividly exemplify the outcomes of these evolutionary trends. Most of the integrating neurons of a mammal are centralized in the brain and spinal cord, and the major integrating organ—the brain—is at the anterior end of the body in the head. Why has cephalization occurred? Most kinds of animals have particularly important sets of sense organs—such as eyes, ears, and sensory tentacles—at their anterior end. This is easy to understand because when an animal moves forward in its environment, its anterior end is the first to make contact with new dangers or opportunities. The concentration of integrating parts of the nervous system at the anterior end facilitates having a concentration of sense organs there.
724
Here we focus on the groups of animals with high degrees of centralization and cephalization. The nervous system in such animals consists of two major parts, the central and peripheral systems. The central nervous system (CNS) consists of relatively large structures that are composed principally of integrating neurons and their associated glial cells. The largest part of the CNS—containing the greatest number of neurons—is the brain.
The CNS functions as a control system, and like all control systems it needs to interact with sensors and effectors to function (see Concept 29.6). The sensors are the sensory cells and sense organs that we’ve just discussed. They provide the CNS with information on the animal’s external environment and internal status. Effectors are cells or tissues that perform actions, that “carry out orders.” Muscle cells are effectors because they can contract and produce motion under orders from the nervous system. Other types of effectors include glands, some of which secrete materials in response to signals from the nervous system, and specialized structures such as fish electric organs (which produce high currents or voltages when ordered to do so by the nervous system).
The peripheral nervous system (PNS) consists of neurons and parts of neurons that are located outside the CNS. These neurons are mostly engaged in two functions related to sensors and effectors. They bring sensory information from sense organs to the CNS. They also carry orders from the CNS to muscle cells or other effectors. For instance, if you touch a hot object with your fingers, neurons in the PNS carry sensory information from temperature sensors in your fingers to the CNS. Other neurons in the PNS carry signals from the CNS to muscles in your arm and fingers, ordering the muscles to contract and pull your fingers away. Nerves, as mentioned before, are bundles of axons grouped together in the PNS, analogous to telephone cables containing many wires.
Neurons are classed as interneurons, sensory neurons, or efferent (including motor) neurons.
- Interneurons are neurons that are confined to the CNS. Most neurons in the CNS are interneurons. For the most part, interneurons engage in integrative and information-storage functions.
- Sensory neurons function as sensory receptor cells or are neurons (also called afferent neurons) that carry signals to the CNS from sensory cells or organs.
- Efferent neurons convey signals from the CNS to muscles or other effectors such as glands. Among these neurons, those that carry signals to skeletal muscles are called motor neurons.
The cellular components of nervous systems—the neurons, synapses, and sensory cells—are remarkably similar in the fundamental ways they function in all groups of animals. However, some animals differ substantially from others in the ways their neurons are organized to form their nervous systems. Let’s briefly compare vertebrates and arthropods to see this point.
In vertebrates, the central nervous system is positioned in the dorsal part of the body (FIGURE 34.21A). It consists of two major parts: the brain in the head and the spinal cord that extends for most of the length of the body behind the brain. Both of these parts are to some degree hollow inside. In short, the vertebrate CNS is dorsal and hollow. The neurons and glial cells in the spinal cord form a continuous column of tissue, in the sense that the cord does not exhibit sharply differentiated subparts along its length.
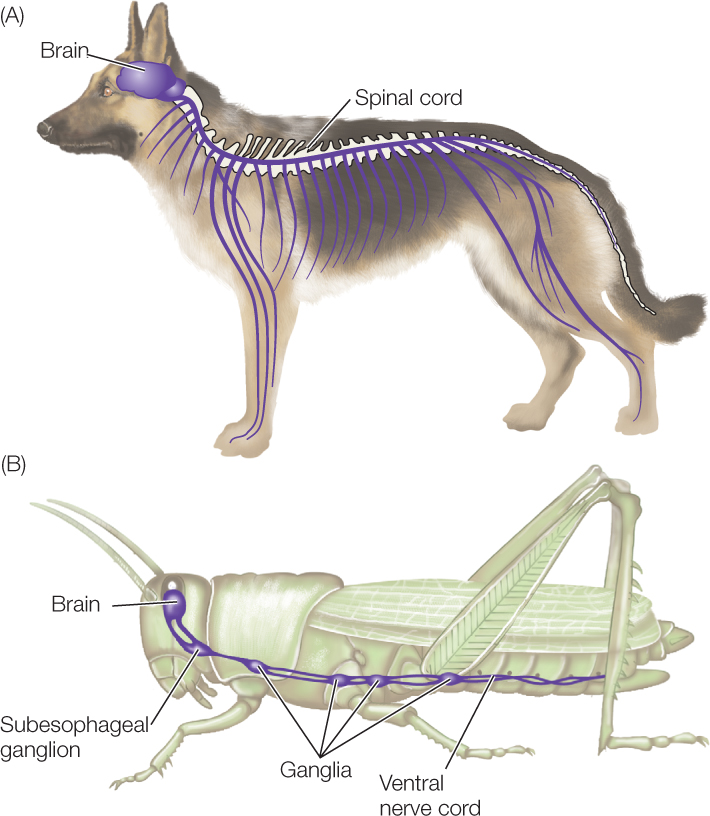
In arthropods, such as insects and crayfish, the CNS is mostly positioned in the ventral part of the body (FIGURE 34.21B; see also Figure 32.4). It too consists of two major parts: the brain, and a ventral nerve cord that extends for most of the length of the body behind the brain. These structures are not hollow. In other words, the arthropod CNS is mostly ventral and solid. Moreover, instead of being a continuous column of tissue, the ventral nerve cord is composed of a series of alternating subparts. Arthropods are segmented (see Concept 23.1), and there is an enlarged structure called a ganglion (plural, ganglia) in each segment of the animal’s body. Two strands of nervous tissue, called connectives, run from each ganglion to the next. (A word of warning: the word “ganglion” is also used in describing vertebrate nervous systems, but it has a very different meaning in discussing vertebrates and arthropods, as we’ll soon see.)
725
The autonomic nervous system controls involuntary functions
The nervous system as a whole controls several types of effectors: skeletal muscles, smooth muscles, glands, and so forth. An important distinction is recognized between the part of the nervous system that controls skeletal muscles and the part that controls all other effectors. This distinction is entirely different from the one between the CNS and PNS.
The autonomic nervous system (ANS) is the part of the nervous system (both CNS and PNS) that controls effectors other than the skeletal muscles. These effectors are called autonomic (or internal) effectors. We generally are unaware that they are being controlled, and we have little or no ability to influence them by conscious, voluntary effort. (“Autonomic” means involuntary or unconscious.) The ANS exists in all groups of vertebrates. Some invertebrates are also considered to have an ANS.
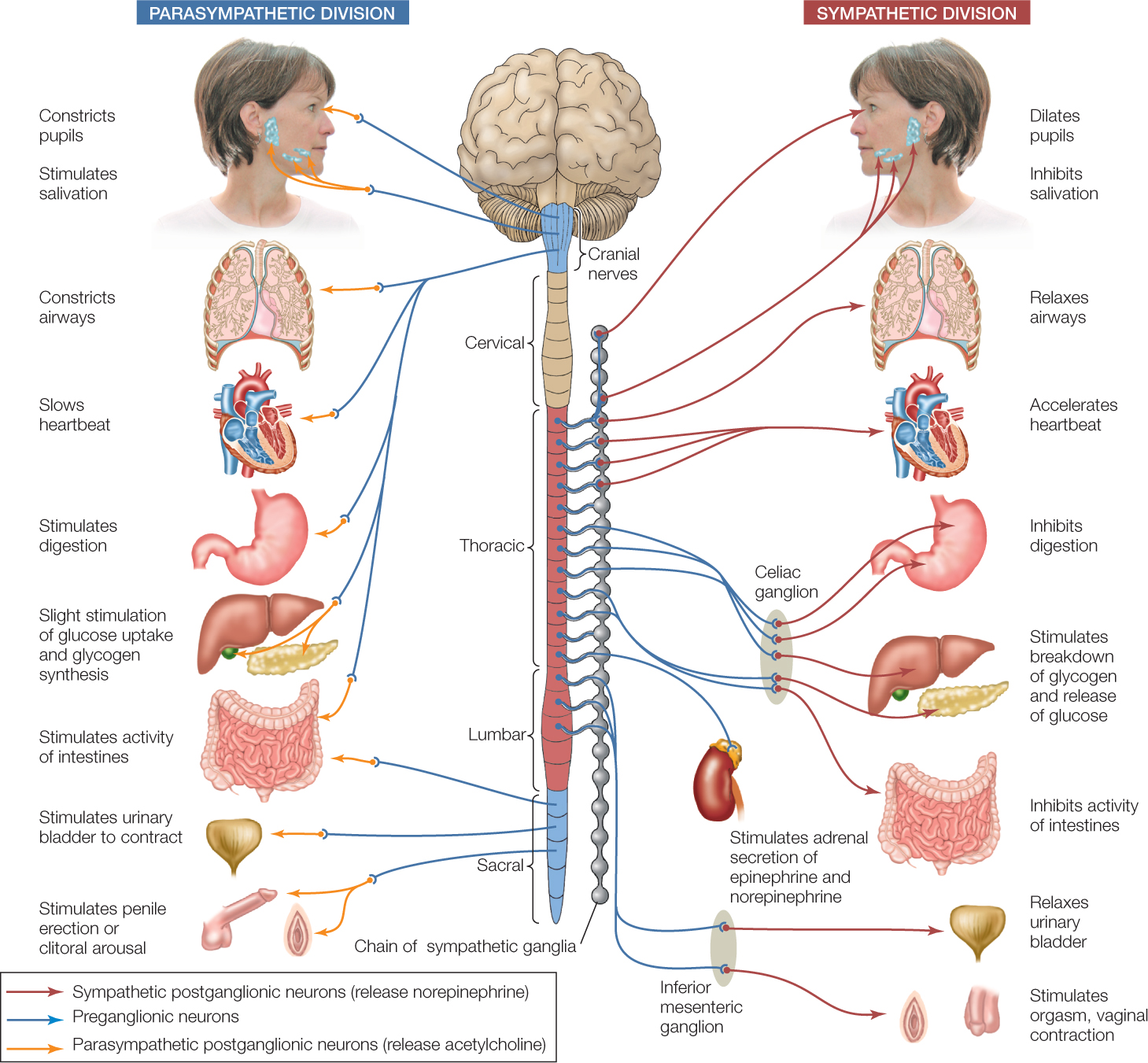
The ANS of a human or other mammal controls a wide variety of smooth muscles, such as those in the gut, blood vessels, bladder, eyes (e.g., iris muscles), airways of the lungs, and reproductive organs (e.g., during orgasm) (FIGURE 34.22). It controls a variety of exocrine glands (glands that secrete their products elsewhere than into the blood) such as the tear and sweat glands. It also controls a few endocrine glands (glands that secrete hormones into the blood), most notably the adrenal medullary glands. And it controls color-change cells in fish, the acid-secreting cells of the stomach, and several other effectors.
726
The vertebrate ANS has three divisions. The enteric division is composed of the nerve cells internal to the gut wall (see Figure 29.14). Here we focus on the sympathetic and parasympathetic divisions (see Figure 34.22).
We can think of these two divisions as consisting mainly of efferent neurons that carry orders from the CNS to autonomic effectors. Each efferent pathway is composed of two neurons that synapse at a ganglion. (As we discuss this topic, remember that “ganglion” has different meanings in vertebrates and arthropods.) A vertebrate ganglion is a discrete, anatomically clustered set of neuron cell bodies in the PNS. The two neurons of each efferent pathway are described as preganglionic and postganglionic.
The preganglionic neurons of the parasympathetic division exit the CNS from two regions: a cranial region where the brain and spinal cord connect and a sacral region, the most posterior region of the spinal cord. The axons of these preganglionic neurons travel almost to their target cells before synapsing with the postganglionic neurons.
In contrast, the preganglionic neurons of the sympathetic division exit the CNS from the thoracic and lumbar regions of the spinal cord. Most of the ganglia of the sympathetic division lie next to the spinal cord, lined up in two chains on either side. Accordingly, the postganglionic neurons need to cover most of the distance to the target cells.
The most important neuronal difference between the sympathetic and parasympathetic divisions is that their postganglionic neurons employ different neurotransmitters where they synapse with target cells. The sympathetic neurons release norepinephrine as their neurotransmitter. The parasympathetic neurons release acetylcholine (ACh). In organs that receive both sympathetic and parasympathetic input, the target cells typically respond in opposite ways to norepinephrine and to ACh.
The sympathetic and parasympathetic divisions thus often work in opposition to each other, as emphasized in Figure 34.22. In this way, the two divisions—acting together—can control autonomic effectors in fine-tuned ways, adjusting their functions up or down as needed. For example, both divisions innervate the pacemaker of the heart. Sympathetic stimulation causes the heart rate to increase, and parasympathetic stimulation causes it to decrease.
The fight-or-flight response is the most famous effect of the sympathetic division. When the sympathetic division is strongly activated, it greatly increases the heart rate, force of heart contraction, and cardiac output. It dilates the passage-ways of the lungs and increases release of glucose into the blood from the liver. In all these ways, it prepares the body to meet an emergency. At the same time, it reduces less urgent activities such as digestion.
Spinal reflexes represent a simple type of skeletal muscle control
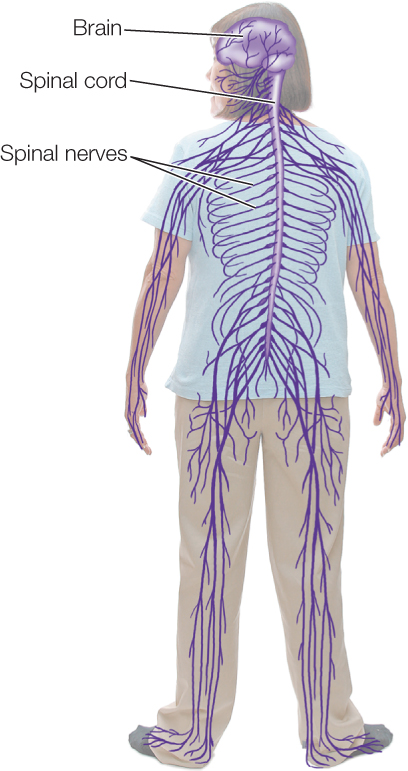
Now let’s turn to control of the skeletal muscles. These effectors are controlled by the parts of the CNS and PNS other than the autonomic nervous system. Many of the neurons that control the skeletal muscles enter or leave the CNS in spinal nerves that connect with the spinal cord at regular intervals on each side (FIGURE 34.23). These nerves contain both motor neurons that carry orders from the CNS to the skeletal muscles and sensory neurons that bring information to the CNS, such as information from stretch receptors in the muscles.
As we all know, we can consciously or voluntarily control our skeletal muscles. This is possible because neurons in our brain can control signals in the motor neurons to the muscles. Also, however, some signals to the muscles originate by way of spinal reflexes. These are neuron-mediated responses that do not involve participation of the brain. Instead, all the neuron interactions occur in the spinal cord, requiring no conscious attention. Because spinal reflex systems are relatively simple, they are among the best-known vertebrate neural circuits.
We are all familiar with the spinal reflex known as the knee-jerk reflex (FIGURE 34.24). It occurs when a physician or nurse taps your knee with a little hammer and—if your nervous system is healthy—your lower leg jerks forward. The tap stretches the patellar tendon that connects your quadriceps muscle—the muscle on the forward-facing side of your thigh—to your tibia bone in your lower leg. The stretch caused by the tap stimulates stretch receptors in the muscle. These receptors initiate action potentials in sensory neurons, which propagate these action potentials along their axons to the spinal cord via a spinal nerve.
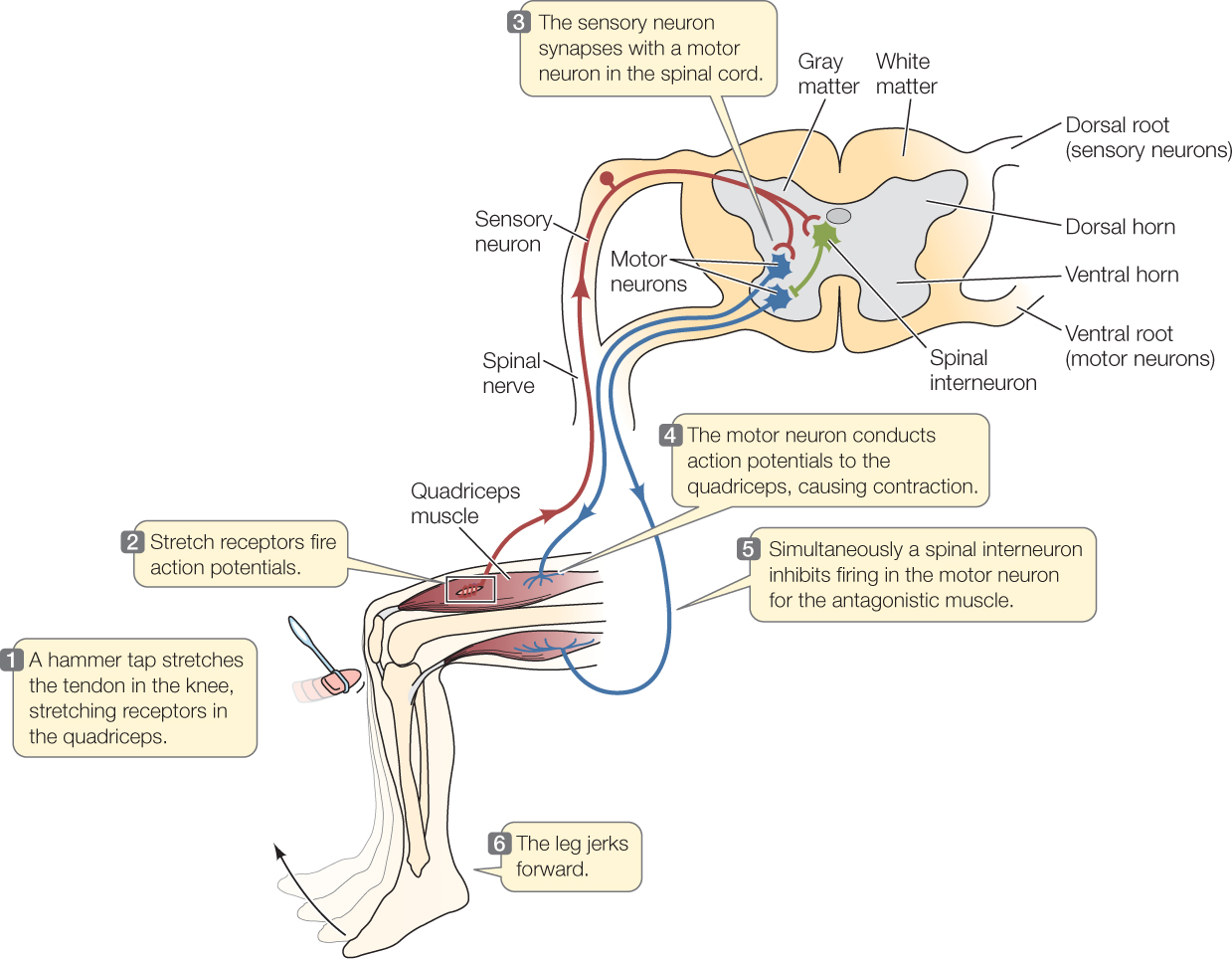

Go to ANIMATED TUTORIAL 34.8 Information Processing in the Spinal Cord
PoL2e.com/at34.8
In the spinal cord, each sensory axon synapses with a motor neuron that serves the same muscle, the quadriceps. This synapse is excitatory and causes the motor neuron to generate action potentials. These signals travel in the axon of the motor neuron via the spinal nerve to the quadriceps. There the action potentials excite the muscle at neuromuscular junctions. The muscle contracts, and your lower leg kicks forward.
727
There’s more to the story. An additional important factor is that limb muscles are organized into opposing (antagonistic) pairs (see Concept 33.2). For contraction of the quadriceps to move your lower leg forward easily, the opposing muscle on the back of your thigh needs to relax. It does so because the sensory neurons coming from the stretch receptors also synapse onto interneurons in the spinal cord (see Figure 34.24, step 5). Those interneurons make inhibitory synapses onto the motor neurons that control the opposing muscle. The motor neurons are inhibited from stimulating that muscle to contract, allowing your lower leg to move forward when the quadriceps contracts.
These interactions among cells are unusually simple, but they nicely illustrate the interaction of sensory and motor neurons, interneurons, and the PNS and CNS in controlling contractions of skeletal muscles.
The most dramatic changes in vertebrate brain evolution have been in the forebrain
All vertebrate brains consist of three major regions: the forebrain (the most anterior part), midbrain, and hindbrain (the most posterior part). The most posterior part of the hindbrain is the medulla oblongata. All information traveling between the brain and spinal cord must pass through the medulla oblongata. During the course of vertebrate brain evolution, the overall shape and relative size of the medulla oblongata have changed little (FIGURE 34.25).
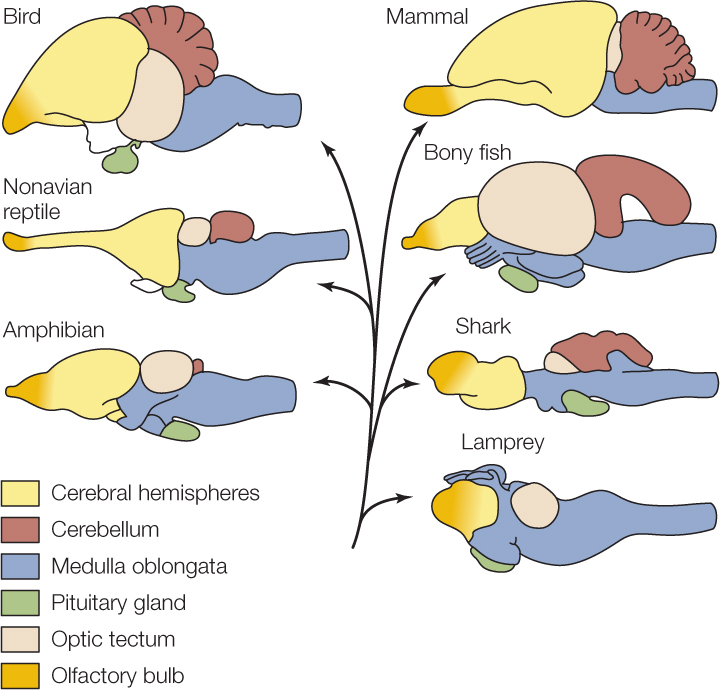
In contrast, the cerebral hemispheres, which are a major component of the forebrain, have undergone dramatic changes during vertebrate evolution. Both mammals and birds have evolved especially large cerebral hemispheres (see Figure 34.25). This is noteworthy because the cerebral hemispheres are particularly important in carrying out high-order sensory, motor, and integrative functions. In humans, for example, the cerebral hemispheres are responsible for language and reasoning.
Mammals and birds also have brains that are about 10 times larger than the brains of lizards or other nonavian reptiles that are the same in body size. A mouse, for example, has a brain about 10 times larger than that of a mouse-sized lizard, and a parrot has a brain about 10 times larger than that of a parrot-sized lizard. The cerebral hemispheres account for much of this difference.
There are two take-home messages from these comparisons. First, mammals and birds have evolved strikingly large cerebral hemispheres, the neurons and glia of which are of enormous importance in high-order brain function. Second, brain size matters. The evolution of enhanced functionality in mammals and birds has gone hand in hand with large increases in the numbers of neurons. From this vantage, it is significant that primates and dolphins have the largest brains, for their body sizes, of all mammals.
728
We need to be careful not to overextrapolate the significance of these broad trends, however. Brain size matters in a general sense, but it does not matter when we look at all particulars. Some animals with small brains exhibit stunning behavioral capacities (see Figure 40.8). Moreover, among human beings—where the subject has been studied for well over a century—all the evidence available indicates that individual intelligence is not correlated with individual brain size. We know too little about brain function to explain how all these pieces of the puzzle fit together.
Location specificity is an important property of the mammalian cerebral hemispheres
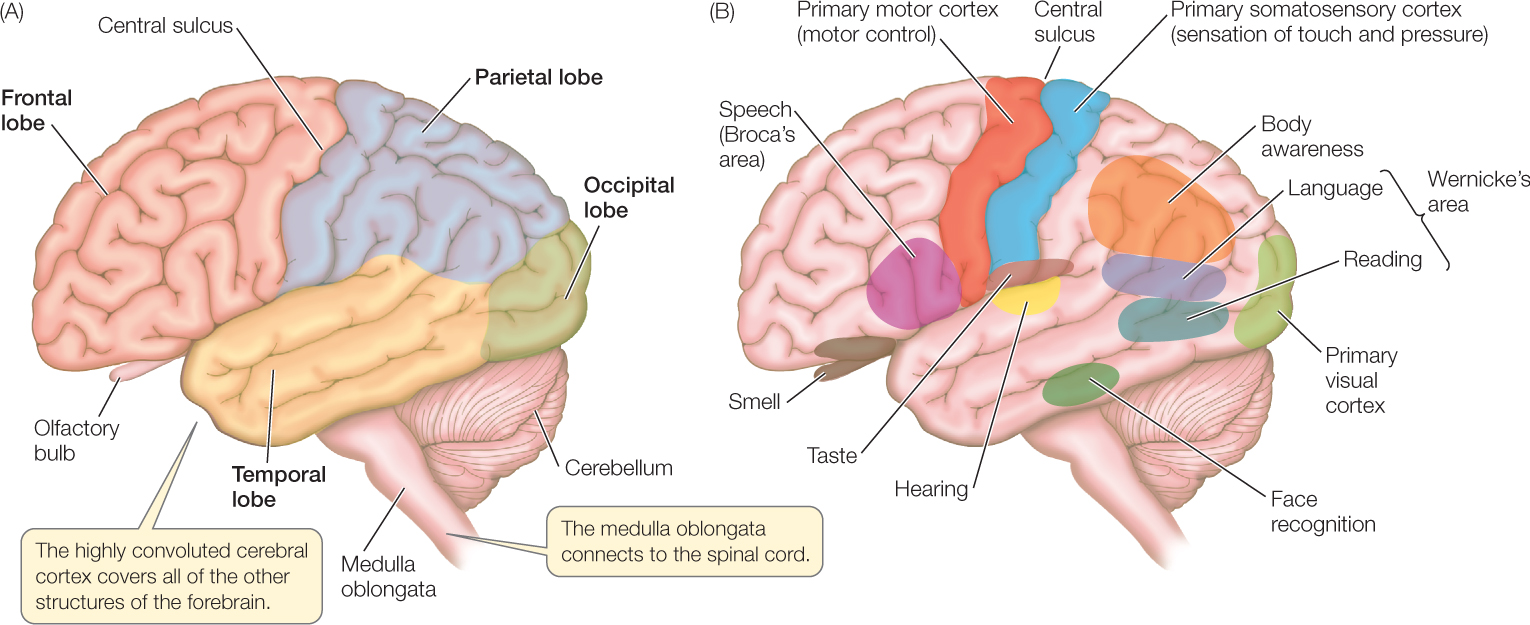
In humans, the cerebral hemispheres (which together make up the cerebrum) are so enlarged that they cover most other parts of the brain. The outermost layer of the cerebral hemispheres is the cerebral cortex, a thin, important layer rich in cell bodies. The cerebral cortex is only about 4 millimeters thick, but it is folded into ridges (convolutions) that increase its size. The cerebral hemispheres play major roles in sensory perception, learning, memory, and conscious behavior. Indeed, they provide the neural resources for the great intellectual capacity of humans.
A curious feature of the mammalian nervous system is that the left side of the body is served (in both sensory and motor aspects) mostly by the right side of the brain, and the right side of the body is served by the left side of the brain. Thus sensory input from your right hand goes to your left cerebral hemisphere, and sensory input from your left hand goes to your right cerebral hemisphere.
Earlier we noted that all action potentials arriving in the brain are the same, even though some represent visual images, others represent sounds, and still others represent odors. Some action potentials from a person’s touch receptors represent touches to the fingers, whereas others represent touches to the abdomen or feet. How does the brain process a simple, invariant electrical event—an action potential—and develop an accurate perception of the type of stimulus (sight, sound, or odor) that caused it, or an accurate perception of the place of origin of the stimulus?
729
Location specificity is one of the major mechanisms the brain uses to interpret the action potentials it receives. In each cerebral hemisphere, various specific regions are specialized in adults to carry out specific sensory and motor functions. Distinct regions are responsible for vision, hearing, and smell, for example (FIGURE 34.26). Axons from the retinas connect to the visual cortex, the region responsible for vision. Similarly, axons from the cochleas in the ears and axons from the odorant receptor cells in the nasal epithelium connect to the regions responsible for hearing and smell, respectively. Because of these physical connections, axons are effectively labeled in regards to the meaning of the action potentials they bring to the brain.
Go to ACTIVITY 34.5 Structures of the Human Brain
PoL2e.com/ac34.5
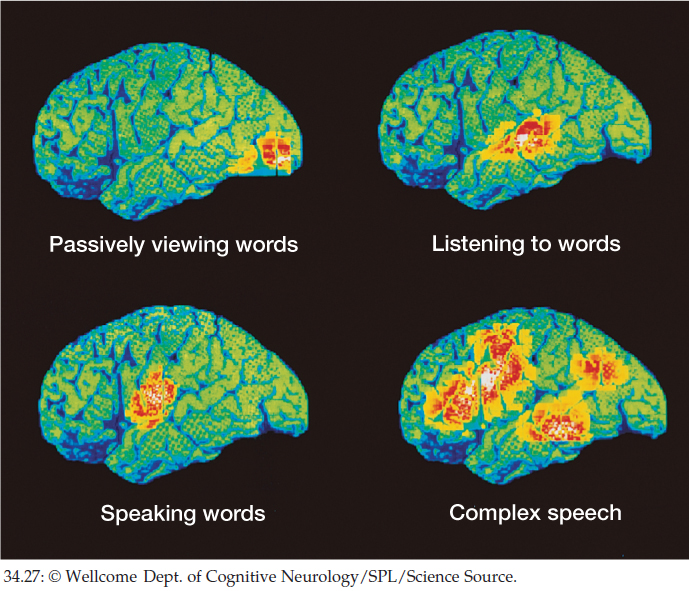
Motor functions are also localized. Moreover, combined sensory and motor functions often occur in certain, relatively localized brain areas. An explosive area of research at present is the exploration of sensory, motor, and combined sensory and motor localization by use of imaging methods that allow us to visualize where neurons exhibit evidence of increased electrical activity. For instance, the positron emission tomography (PET) images in FIGURE 34.27 show brain regions where there is evidence of increased neuronal activity when several language functions are carried out. Functional magnetic resonance imaging (fMRI) is another technique used for similar research.
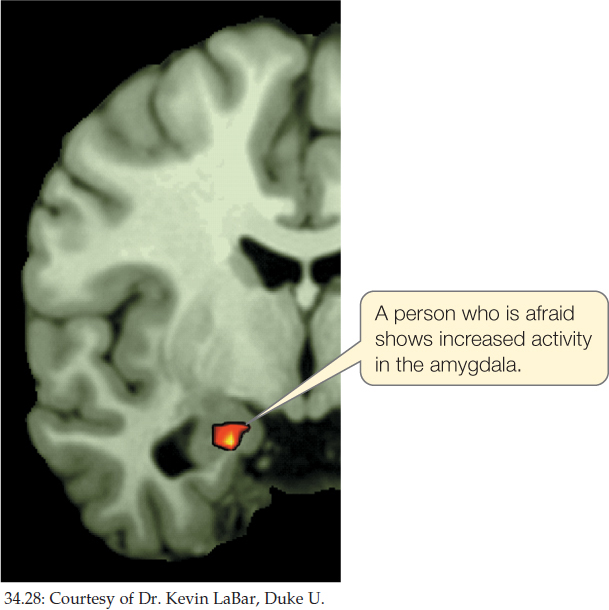
Localization of function is not limited to the cerebral hemispheres. FIGURE 34.28, for example, is a fMRI image of the brain of a person experiencing fear. The image indicates increased neuronal activity in the amygdala, a part of the forebrain (outside the cerebral hemispheres) that is well known to participate in fear responses. Abundant evidence points to the hippocampus, still another part of the forebrain, as being critical for spatial learning and memory (we discussed synaptic plasticity in the hippocampus in Concept 34.3). Damage to the hippocampus renders mice and rats unable to solve maze problems that depend on learning the relationships of objects in space.
Brain regions have maps in some cases. In such regions, the parts of the brain that serve various anatomical regions of the body are physically related to each other in ways that mirror the physical relationships of the rest of the body. A well-known example is seen in the somatosensory (“body sensing”) part of the cerebral cortex. Regions of the cerebral cortex that process sensory information from touch and pressure receptors on the body surface in the leg, hip, trunk, neck, and head are lined up (FIGURE 34.29A). Similarly, regions processing such information from the eyes, nose, lips, and jaw are lined up. In the illustration, the size of each body part in the drawing reflects the amount of cortical area devoted to the part. The large size of the lips, for example, reflects the fact that a large area is devoted to processing sensory signals from the lips.
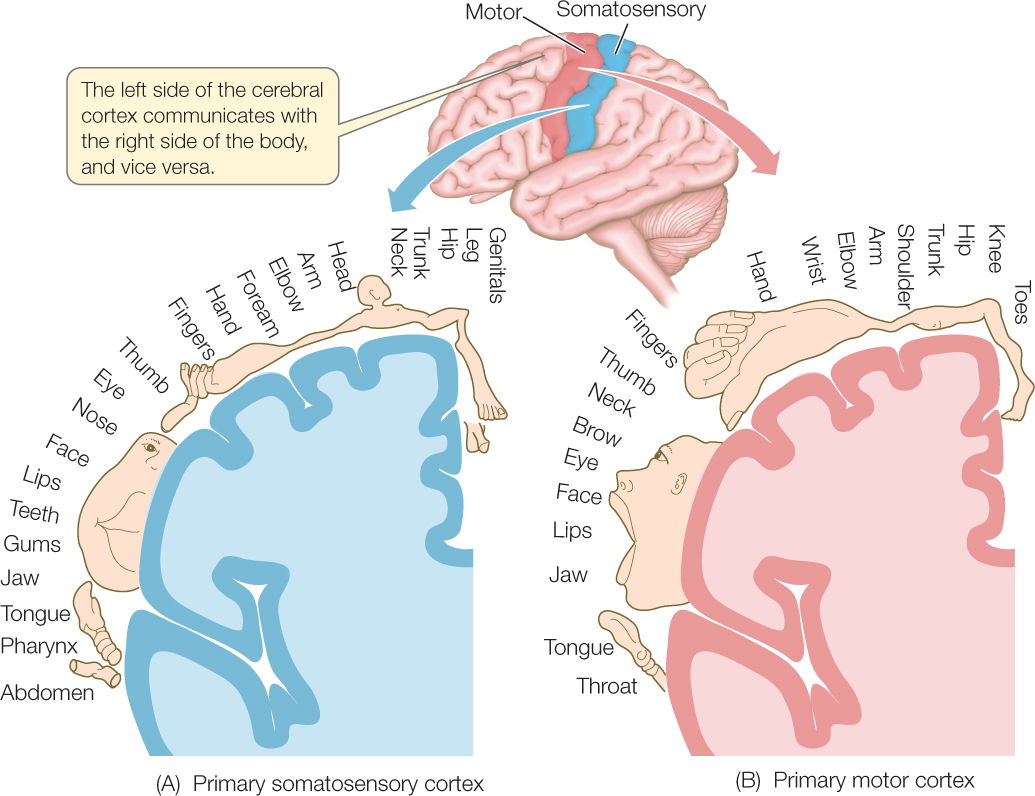
Near the mapped somatosensory area of the cortex is a parallel, mapped motor area. You can see from the size of drawings of body parts in the motor area (FIGURE 34.29B) that large areas of motor cortex are devoted to sending motor signals to the hands and face.
Not all parts of the brain fit easily into the concepts of localized function and mapped brain regions. Thus we need to recognize that these concepts are both important and limited in their ability to clarify brain function.
A curious aspect of language processing in humans is that it resides principally in one cerebral hemisphere—which in about 85 percent of people is the left hemisphere. The two hemispheres share responsibilities for other functions. Individuals with damage to the left hemisphere frequently experience deficits in their ability to use or understand words. The deficit depends on the specific part of the left hemisphere that is damaged. Damage to Broca’s area, for example, interferes with speech and writing, whereas damage to Wernicke’s area interferes with understanding language (see Figure 34.26B).
730
The development of a full understanding of the brain is one of the great frontiers of twenty-first century biology. Many major unanswered questions remain. “What is consciousness?” is one of the most compelling.
CHECKpoint CONCEPT 34.5
- Give two specific examples in which the sympathetic and parasympathetic divisions exert opposite effects on the function of an organ.
- Why is localization of function in the brain critical for sensory perception?
- What is cephalization of the nervous system, and why has it evolved in so many animals? Try to think this through using the knowledge you have gained about the relationships between the brain and sense organs.
Question 34.2
How might the star-nosed mole’s brain be specialized to process information from its nose?
ANSWER Just as large areas of cerebral cortex are devoted to processing sensory information from a person’s lips and fingers (see Figure 34.29A), large areas of a star-nosed mole’s cerebral cortex are devoted to processing sensory information from the tentacles of its nose. The drawings in FIGURE 34.30A show a mole in two ways: first, proportioned as it actually is and, second, proportioned in a way that reflects how much cortical tissue is devoted to processing sensory information from its specific body parts, such as its nose, eyes, whiskers, and anterior digging feet. Note that the amount of cortical tissue devoted to processing signals from the nose is larger than that devoted to the rest of the mole’s body.
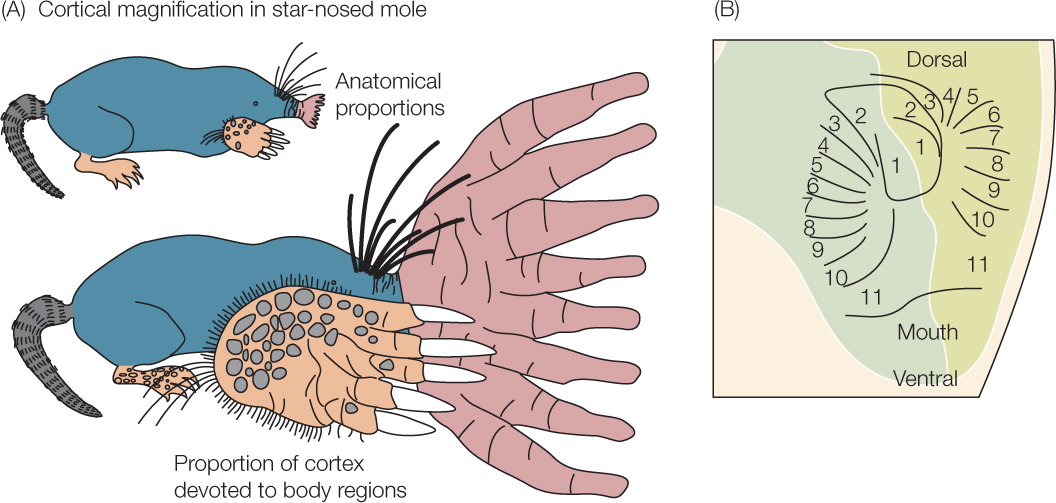
Additionally, a localized part of the cerebral cortex is devoted to receiving sensory information from the nose, and this part of the cortex is highly mapped. The tentacles surrounding each nostril are numbered from 1 to 11 in order from the most dorsal-pointing tentacle to the most ventral-pointing one. FIGURE 34.30B shows the relevant part of the cortex, including the places in the cortex that receive axons from the numbered tentacles. Note that the map of the cortical regions exactly mirrors the arrangement of the tentacles. By means of such localization in the cortex, the brain can readily determine the tentacle that originates any action potential that arrives by way of any particular axon from the nose.
The concepts of localized function and mapped brain regions apply to the star-nosed mole as much as they do to people, reflecting the fact that the mole’s entire nervous system—including its sense organs—is highly integrated.
731